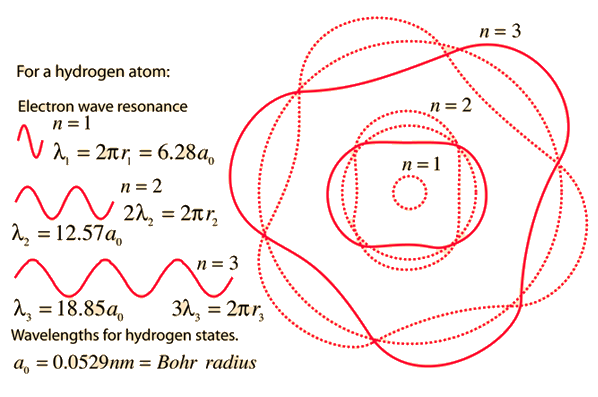
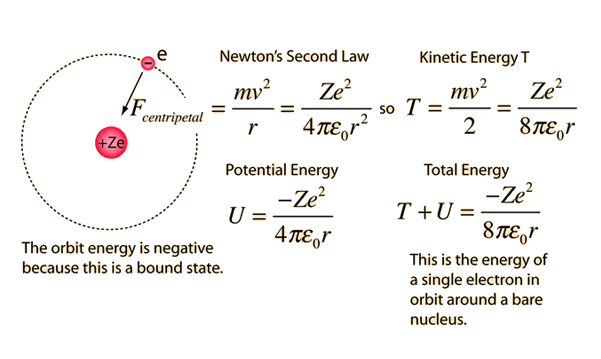
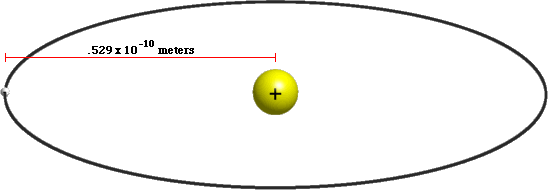
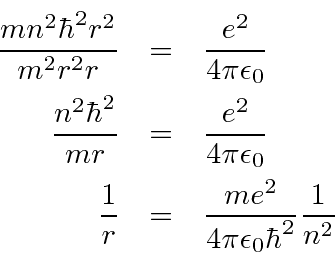MathType - The most probable distance between the nucleus and the electron in a hydrogen atom in its ground state is given by the Bohr Radius. This physical constant is named after

Physics - Ch 66.5 Quantum Mechanics: The Hydrogen Atom (6 of 78) How was the Bohr Radius Determined2 - YouTube
Calculate radius of second Bohr radius of hydrogen atom and then also calculate the speed of the electron and total - Sarthaks eConnect | Largest Online Education Community

Question Video: Understanding the Effects of Electron Mass on Orbital Radius Using the Bohr Model | Nagwa
What is the most possible radius (in PM) for an electron in the first orbit of a hydrogen atom? - Quora

Radius of nth Bohr's orbit-Equation -Shortcut formula-Solved problems-IIT JEE NEET GATE Adichemistry - YouTube

PPT - 1. The first Bohr's radius (for electron in hydrogen atom in the ground state): PowerPoint Presentation - ID:6524739