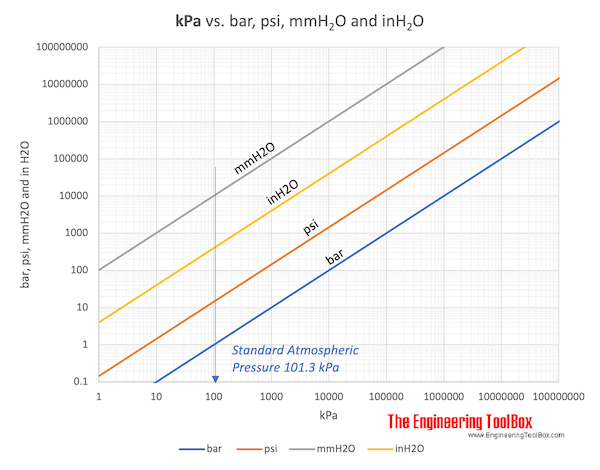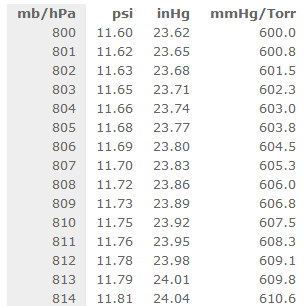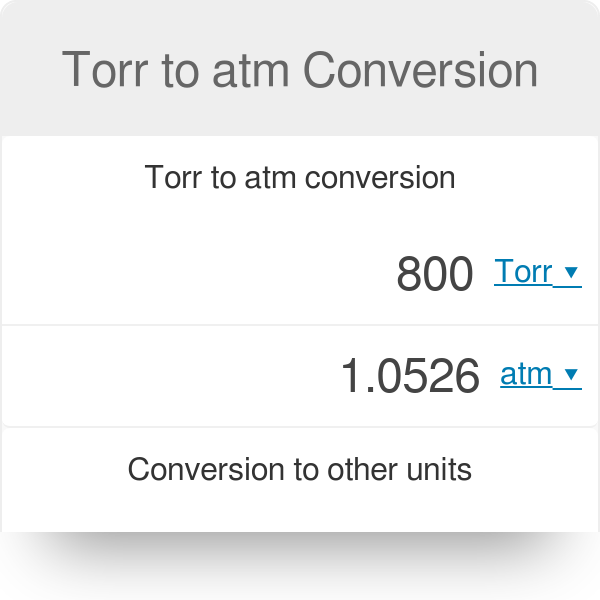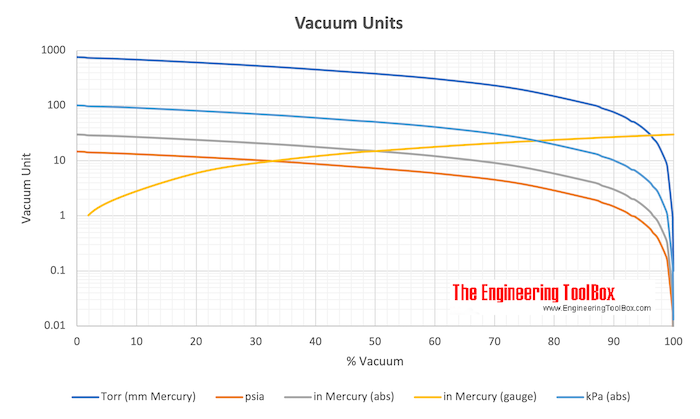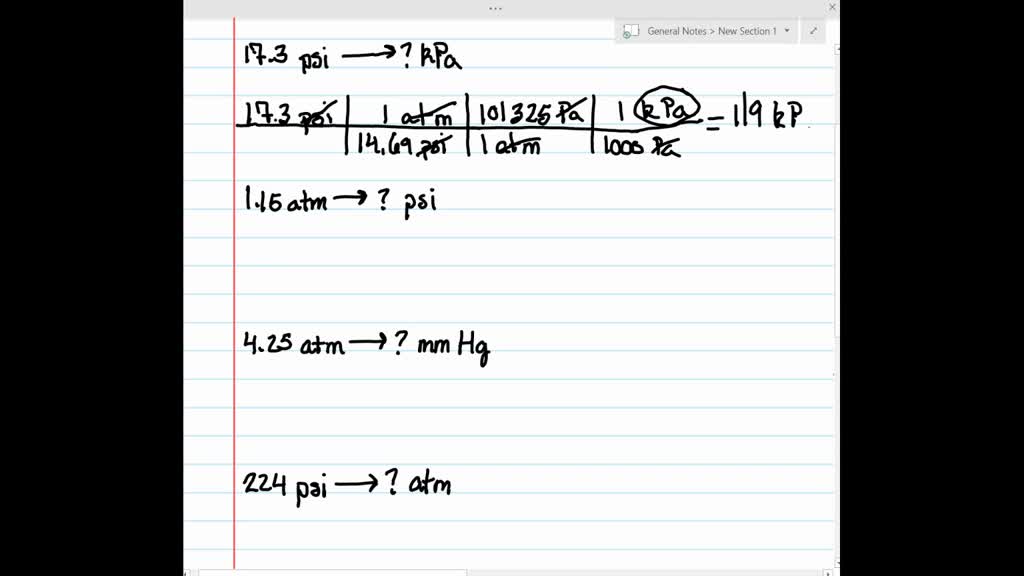
SOLVED:Make the indicated pressure conversions. a. 17.3 psi to kilopascals b. 1.15 atm to psi c. 4.25 atm to mm Hg d. 224 psi to atmospheres

SOLVED:The pressure on top of Mount Everest (29,029 ft or 8848 m) averages about 235 mmHg. Convert this pressure to \begin{equation}\begin{array}{ll}{\text { a. torr }} & {\text { b. psi }} \\ {\